Using Excel to track Food product Quality Control
- Maxence Duprez
At the beginning of Food Quality Control
Quality to develop your company
Relievd* is a new brand of dietary supplements founded in late 2019. Relievd specifically targets young retirees looking to maintain an active lifestyle. Present primarily online and increasingly in pharmacy networks, Relievd has built its growth on the quality of its products and transparency with its educated and demanding customers.
Relievd offers a range of 10 products and aims to complete it with the launch of 10 new formulas within 2 years. Composed of a team of 15 people, Relievd subcontracts its entire production to 3 manufacturers of food supplements depending on the type of galenic. The manufacturers take care of the purchase of raw materials and packaging, except for some specific ingredients that are purchased directly by Relievd. The quality control of the products is ensured by the development and formulation team. With each order, the processors send a certificate of analysis for the received batch and Relievd simply checks the conformity.
As sales grow, brand visibility increases and the range expands, Relievd has deployed ERP (enterprise resource planning) software to track stock levels, customer orders and invoicing. While it is possible to give a quality status and release a product, the ERP does not allow for accurate quality control.
Relievd wants to implement a real quality control system to ensure the traceability of its products and guarantee the performance of its formulas.
It is therefore decided to:
- Define precise specifications for directly purchased ingredients and finished products.
- To systematically recover the certificates of analysis of the batches received and to record the results.
- To set up a system of periodic analyses to control the results of the suppliers of ingredients and manufacturers of food supplements.
Right click, save as…
To do this, without any necessary investment, the first and quickest solution is to use classic office tools, spreadsheets, folders & subfolders.
The first step is to manage the quality documents with folders and subfolders. Main folders according to the type of products, then, on one side, a sub-folder per product with the specification sheet and the quality information. On the other side, another sub-folder with the certificates of analysis which will be sorted by date of addition or by name. It is obviously possible to refine and complicate the tree structure. At each new reception, it is sufficient to save the certificates of analysis of the suppliers and laboratories in the sub-folders by updating the name of the file to be able to find them easily.
Self-checking strategy
The next step is to analyze the materials and finished products to check the suppliers’ results. If all the analyses cannot be launched at once, the quality manager is aware of the most critical materials and products that must be analyzed in priority. As there are still few products and analyses to be performed, he also knows the laboratories to outsource them to.
To compile the analytical results, a spreadsheet will do the trick. We start by entering the specifications of the products. When a new batch is received, these specifications are copied and pasted, then in additional columns, the batch number and the result per analysis are indicated in order to validate the conformity. If you need to edit a certificate of analysis, you can use predefined templates to save the values in a final file.
For young companies with about twenty products in their catalog, 5 to 10 batches or about ten analyses per month, this is a system that works very well. Economical and practical, it allows to start organizing information and to set up a quality system without constraints.
Excel for quality control, almost done ?
However, as soon as you increase the number of references, batches and analyses, the system becomes more complex and spreadsheets and files show their limits. It is necessary to be very rigorous in the naming and filing of files to find them easily. Creating test order forms and managing sample shipments becomes tedious. Issuing new certificates of analysis, validating them and registering them is becoming increasingly time-consuming. Finally, to avoid the explosion of analytical costs, periodic analysis plans are increasingly used. Unfortunately, due to the lack of consolidated data, this is often done in an empirical way without any real justification. Excel for quality control seems to be inefficient.
It is then time to think about the next step to structure something else than an Excel quality control system. Relieved operates as a subcontractor, both for the production of its products and for their analysis. Therefore, they do not need a real laboratory management software (LIMS) which are more made to manage a park of analytical machines or the schedules of the laboratory technicians.
The sustainability of the quality system
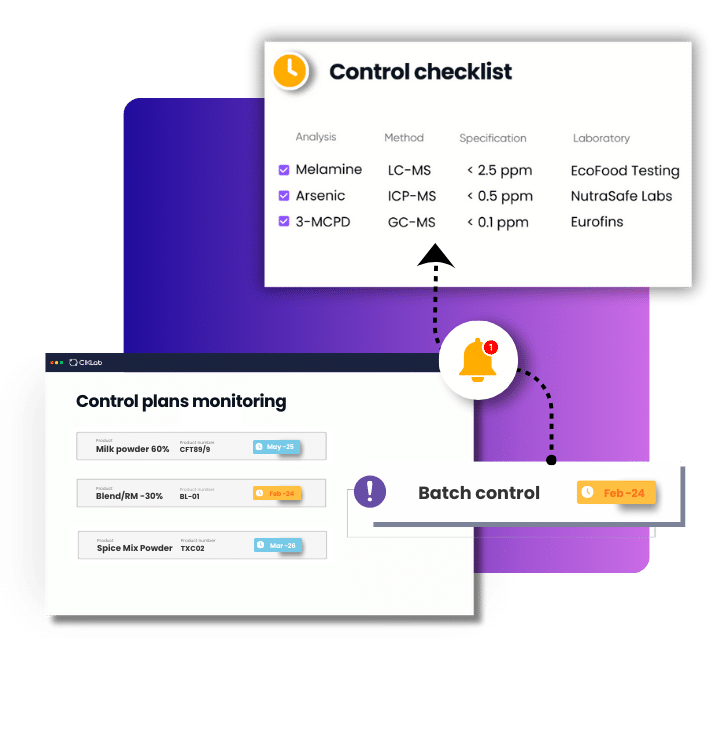
In this case CIKLab is a simple alternative, adapted to Relievd’s needs. First of all, CIKLab allows the quality manager to create analyses, specification sheets and batch sheets. All supplier information is centralized and there is no more file tree to set up like it is for Excel for quality control.
Then, CIKLab makes it possible to link product control analyses to different analysis laboratories according to their expertise. When a new batch is received, it is very easy to see which analyses need to be launched and it only takes one click to edit the analysis order forms. CIKLab thus ensures the sustainability of the quality system.
Analysis results from laboratories and even suppliers are entered into CIKLab in order to validate product conformity. It is also possible to extract and compare analysis results over a given period. This allows to compare suppliers and laboratories results and to statistically validate the consistency of products to adjust the periodicity of the control plans accordingly.
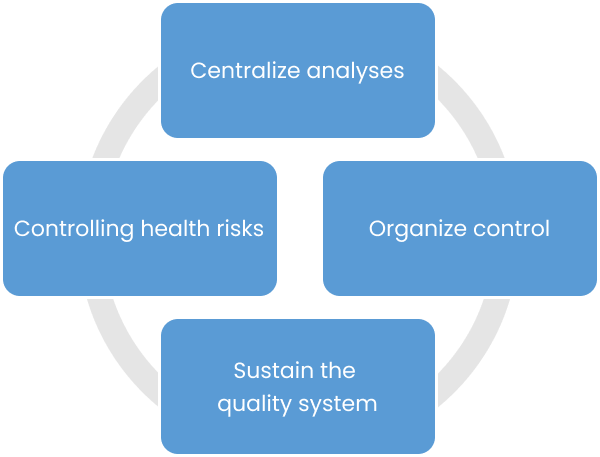
Using CIKLab allows you to structure your quality control and save time in document management; to be more efficient in ordering and monitoring subcontracted analyses; to build and exploit your analytical assets to adjust your control plans and limit your control costs.
Why waiting?
But then, why wait to structure your quality system? The cost of quality will always be lower than the cost of non-quality.
Even if you only perform a few analyses at the beginning, creating product sheets and recording supplier data by batch allows you to build your analytical assets. Excel for quality control works for this time. This is an interesting first indicator to follow the evolutions over time and allows you to build your quality system progressively as your product portfolio develops.
You work in food quality control and you’re still using Excel to manage all your...