Reliable contract laboratory services for Food Safety: A strategic quality assurance partnership
- Lionel MURAT
In the food industry, reliable analytical results are a critical cornerstone of food safety assurance. Selecting a competent contract laboratory is not straightforward—it’s a strategic requirement, particularly for quality control departments. The key lies in understanding which criteria to verify to ensure result reliability and product compliance.
In the food industry, food safety relies on an essential foundation of quality control services: the reliability of analytical results. Whether tests are performed in-house or entrusted to a contract laboratory or analytical service provider, their competence and compliance directly determine the quality and safety of products brought to market.
The FSSC 22000 V6 standard, particularly requirement 2.5.1 a) (Management of services and purchased materials), emphasizes the necessity of ensuring that external service providers, including laboratories, meet strict competence and compliance criteria.
“When analyses of parameters critical to food safety are outsourced, the organization must ensure that the laboratory used operates in accordance with applicable ISO/IEC 17025 requirements.”
In other words, while the standard doesn’t mandate that laboratories must be ISO 17025 accredited, it nevertheless requires that their methods and operations comply with these quality standards.
What is ISO/IEC 17025? Why is it important for every laboratory?
The ISO/IEC 17025 standard defines general requirements specifically concerning the competence of calibration and testing laboratories.
It covers several key areas:
- Impartiality and confidentiality
- Organization and resource management
- Personnel qualification
- Metrological traceability of equipment and external supplier management
- Process monitoring including method validation
- Sample management and measurement uncertainty control
- A management system to ensure continuous improvement and result reliability
By complying with this standard (and being accredited), a laboratory demonstrates its ability to produce valid and reliable results, thereby strengthening client and partner confidence and facilitating international recognition of analytical results.
Even if your contract laboratory isn’t formally accredited, working with a partner aligned with ISO 17025 requirements is a reliability guarantee. However, you’ll need to verify several key points as indicated by FSSC 22000 2.5.1 a).
For external laboratories that are not ISO/IEC 17025 accredited, or whose accreditation doesn’t include required test methods, the service level agreement must define at least for these non-accredited methods:
- Test parameters
- Agreed test methods (including their verification and validation)
- Sampling protocol
- Successful participation in proficiency testing programs approved by regulatory authorities
What is laboratory accreditation?
ISO/IEC 17025 accreditation isn’t simply a marketing label for laboratories: it’s formal recognition by an accreditation body (in France, COFRAC) of a laboratory’s technical competence to provide reliable and impartial results.
Key elements include:
- Competence: Personnel are qualified
- Impartiality: Laboratory remains neutral and reliable
- Equipment: Facilities and maintenance are demonstrated
- Traceability: Results linked to SI or reliable reference
- Methods: Analyses and procedures validated and compliant with normative and regulatory requirements
- Measurement uncertainty: Evaluated and communicated
- Validity: Results verified and reliable
- Information system: Data controlled and secured
- Quality management: Effective and integrated system
- Risks/opportunities: Must be considered in management
All these elements ensure a very high level of confidence in obtained results!
International recognition of ISO 17025: The ILAC agreement
A key aspect of ISO/IEC 17025 accreditation is its worldwide recognition through the ILAC MRA (International Laboratory Accreditation Cooperation – Mutual Recognition Arrangement). This multilateral agreement links accreditation bodies from numerous countries (including COFRAC in France) and guarantees that:
- Results issued by an accredited laboratory in a signatory country are accepted without requiring new testing
- Clients can have confidence in the comparability and integrity of analytical results, even when used abroad.
Practically, for an exporting food company, working with an ISO/IEC 17025 accredited laboratory by an ILAC MRA member organization means their analytical reports will have evidentiary value in most international markets.
Outsourcing analyses: A strategic choice for food quality control
Entrusting food testing to specialized contract laboratories has long been established as an essential approach in the food industry.
This strategy offers advantages:
- Access to cutting-edge methods and equipment
- Human, technical, and regulatory expertise
- Guarantee of reliable results compliant with current standards
Facing the growing complexity of food controls (microbiology, chemical contaminants, allergens, authenticity), outsourcing enables meeting regulatory requirements and client/consumer expectations increasingly concerned with transparency and safety!
That said, externalizing analyses also engages your responsibility: you remain accountable for results in the eyes of clients and authorities.
FSSC 22000 2.5.1 requirements applied to laboratories
FSSC 22000 is now one of the most recognized standards for food safety management systems. Based on ISO 22000 and enriched with additional requirements, it establishes a rigorous framework to ensure hazard control and reliability throughout the entire control chain. In version 6, chapter 2.5.1 “Management of services and purchased materials” presents a key point: external suppliers and service providers, including contract laboratories, must be considered true quality partners and demonstrate their competence.
In practice, this means laboratory selection isn’t a simple administrative act, but a rigorous, documented qualification process integrated into the food safety management system.
As a manufacturer, you must consider your contract laboratories as true partners, as they’re part of your quality control service. FSSC 22000 v6, in chapter 2.5.1, specifies that:
“The organization must ensure that purchased products and services conform to specified requirements. When monitoring or measurement activities are outsourced, they must be performed by a service provider demonstrating competence, for example through accreditation.”
This means laboratory selection isn’t an “administrative” act but a rigorous qualification process that must be documented in your food safety management system.
Implementing internal selection and monitoring procedures
To secure your control chain, it’s recommended to:
- Evaluate your laboratory before contracting: accreditation and scope, comparative testing, possibly audits
- Maintain an updated supplier file containing competence evidence (certificates, methods, reports)
- Monitor performance: deadline compliance, technical support, communication, comparative testing
- Conduct regular audits to verify continued compliance with requirements
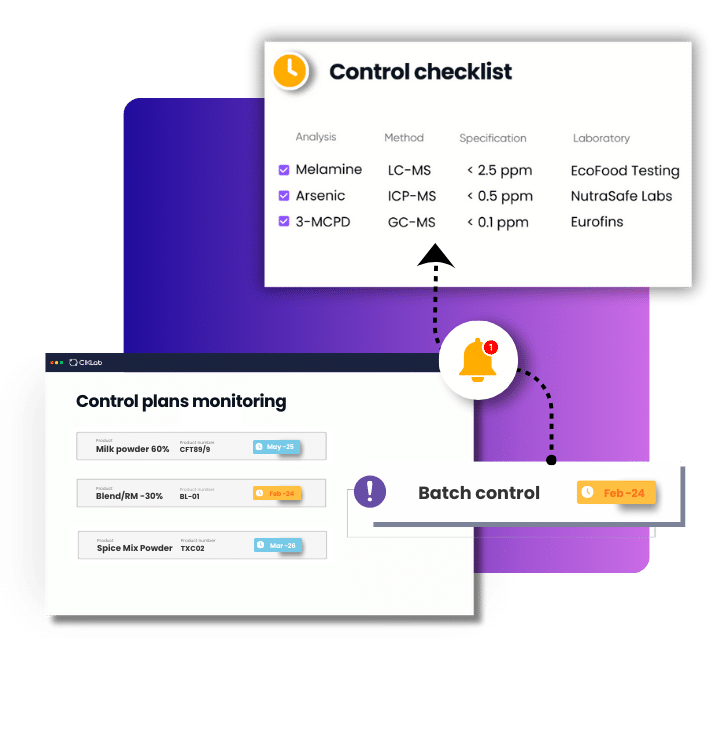
How CIKLab software helps manage your contract laboratories
In the food industry, hazard exposure is constant, and hazards and contaminants to control are numerous: microbiology, micropollutants, natural contaminants, chemical contaminants, etc.
It’s absolutely necessary to have one or more contractors capable of covering all analyses and especially a centralized management system to manage all analytical expertise.
Quality control software like CIKLab enables:
- Managing numerous contractors
- Centralizing all analyses, methods, and prices for each laboratory
- Tracing all analyses and results from raw materials to finished products
- Automatically storing and classifying all test reports from contractors
- Quickly comparing all your laboratories
Conclusions
Food safety tolerates no approximation! Choosing a competent contract laboratory isn’t just a regulatory obligation: it’s a strategic element of your quality control management!
By relying on:
- FSSC 22000 2.5.1 requirements
- ISO/IEC 17025 principles
- Appropriate management tools like CIKLab
…you can guarantee that your analyses, whether internal or outsourced, will be managed very easily!
You work in food quality control and you’re still using Excel to manage all your...